
The process of a liquid becoming a gas is called boiling (or vapourization), while the process of a gas becoming a liquid is called condensation. At a certain temperature, the particles in a liquid have enough energy to become a gas. The atoms in a solid are tightly bound to each other, either in a regular geometric lattice (crystalline solids, which include metals and ordinary water ice) or irregularly (an amorphous solid such as common window glass). The phase change between a liquid and a gas has some similarities to the phase change between a solid and a liquid. Image caption, Scanning electron micrograph of a pre-solar grain. Liquid: Assumes the shape of its container and has a definite volume. The atoms in almost all solids are arranged in regular patterns, called crystals. It has form, hardness or rigidity, or the ability to oppose a change of shape. 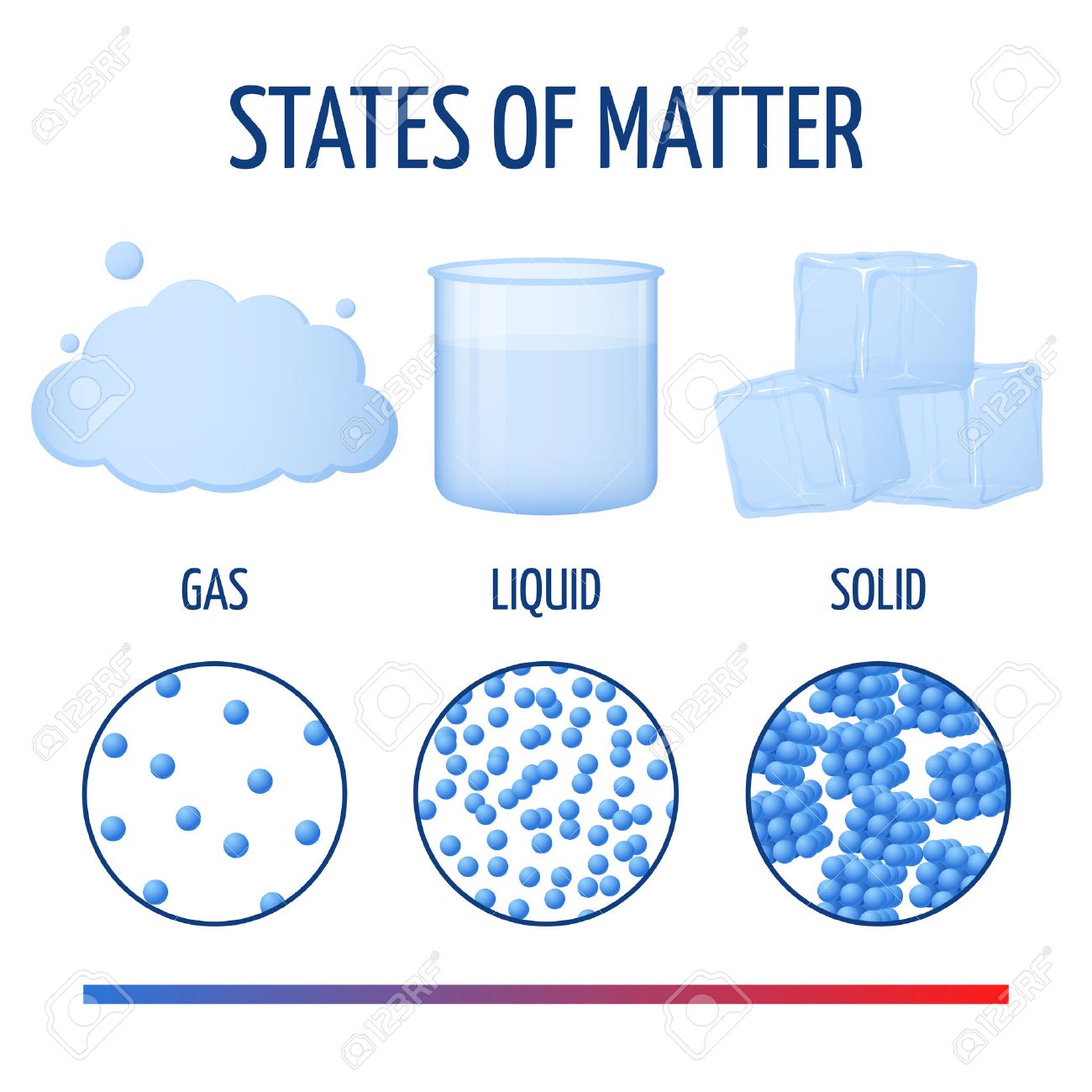
Unlike a liquid, a solid object does not flow to take on the shape of its container, nor does expands to fill the entire volume available to it like a gas. These rays are high-energy particles that travel through our galaxy and penetrate solid matter. Solid: Has definite shape and volume and has the greatest amount of mass per unit of volume or density. Solid are characterized by structural rigidity and resistance to changes of shape or volume.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
